(Whole Blood/Serum/Plasma) Package Insert English A rapid test for a qualitative test for the detection of NS1 antigen, IgG and IgM antibodies of dengue virus in human whole blood, serum or plasma. For professional in vitro diagnostic use only. INTENDED USE The Dengue Combo Test Cassette (Whole Blood/Serum/Plasma) is a rapid chromatographic immunoassay for the qualitative detection of NS1 antigen and IgG and IgM antibodies of Dengue virus in human whole blood, serum, or plasma as an aid in the diagnosis of Dengue infections. SUMMARY Dengue is a flavivirus, transmitted by Aedes aegypti and Aedes albopictus mosquitoes. It is widely distributed throughout the tropical and subtropical areas of the world,1 and causes up to 100 million infections annually.2 Classic Dengue infection is characterized by a sudden onset of fever, intense headache, myalgia, arthralgia and rash. Primary Dengue infection causes IgM antibodies to increase to a detectable level in 3 to 5 days after the onset of fever. IgM antibodies generally persist for 30 to 90 days.3 Most Dengue patients in endemic regions have secondary infections,4 resulting in high levels of specific IgG antibodies prior to or simultaneous with IgM response.5 Therefore, the detection of specific anti-Dengue IgM and IgG antibodies can also help to distinguish between primary and secondary infections. NS1 is one of 7 Dengue Virus non-structural proteins which are thought to be involved in viral replication. NS1 exists as a monomer in its immature form but is rapidly processed in the endoplasmic reticulum to form a stable dimer. A small amount of NS1 remains associated with intracellular organelles where it is thought to be involved in viral replication. The rest of NS1 is found either associated with the plasma membrane or secreted as a soluble hexadimer. NS1 is essential for viral viability but its precise biological function is unknown. Antibodies raised in response to NS1 in viral infection can cross react with cell surface antigens on epithelial cells and platelets and this has been implicated in the development of Dengue Hemorrhagic fever. The Dengue IgG/IgM Rapid Test Cassette (Whole Blood/Serum/Plasma) is a rapid test that utilizes a combination of Dengue antigen coated colored particles for the detection of IgG and IgM Dengue antibodies in human whole blood, serum, or plasma. The Dengue NS1 Rapid Test Cassette (Whole Blood/Serum/Plasma) is a rapid test that utilizes a combination of Dengue antibodies coated colored particles for the detection of Dengue NS1 antigen in human whole blood, serum, or plasma. PRINCIPLE The Dengue IgG/IgM Rapid Test Cassette (Whole Blood/Serum/Plasma) is a qualitative membrane- based immunoassay for the detection of Dengue antibodies in whole blood, serum, or plasma. This test consists of two components, an IgG component and an IgM component. In the IgG component, anti-human IgG is coated in IgG test line region. During testing, the specimen reacts with Dengue antigen-coated particles in the test cassette. The mixture then migrates upward on the membrane chromatographically by capillary action and reacts with the anti-human IgG in IgG test line region. If the specimen contains IgG antibodies to Dengue, a colored line will appear in IgG test line region. In the IgM component, anti-human IgM is coated in IgM test line region. During testing, the specimen reacts with anti-human IgM. Dengue IgM antibodies, if present in the specimen, reacts with the anti- human IgM and the Dengue antigen-coated particles in the test cassette, and this complex is captured by the anti-human IgM, forming a colored line in IgM test line region. Therefore, if the specimen contains Dengue IgG antibodies, a colored line will appear in IgG test line region. If the specimen contains Dengue IgM antibodies, a colored line will appear in IgM test line region. If the specimen does not contain Dengue antibodies, no colored line will appear in either of the test line regions, indicating a negative result. To serve as a procedural control, a colored line will always appear in the control line region, indicating that the proper volume of specimen has been added and membrane wicking has occurred. The Dengue NS1 Rapid Test Cassette (Whole Blood/Serum/Plasma) is a qualitative membrane- based immunoassay for the detection of Dengue NS1 antigen in whole blood, serum, or plasma. During testing, the specimen reacts with Dengue antibody-conjugate in the test cassette. The Gold antibody conjugate will bind to Dengue antigen in the specimen sample which in turn will bind with Anti-Dengue NS1 coated on the membrane. As the reagent moves across the membrane, the Dengue NS1 antibody on the membrane will bind the antibody-antigen complex causing pale or dark pink line to form at the test line region of the test membrane. The intensity of the lines will vary depending upon the amount of antigen present in the sample. The appearance of pink line in the test region should be considered as positive result. REAGENTS The Dengue IgG/IgM Rapid Test Cassette contains Dengue antigen conjugated gold colloid particles, anti-human IgM, anti-human IgG coated on the membrane. The Dengue NS1 Rapid Test Cassette contains anti-Dengue Ag conjugated colloid particles, anti- Dengue Ag coated on the membrane. PRECAUTIONS |
|
| • | For professional in vitro diagnostic use only. Do not use after expiration date. |
| • | Do not eat, drink or smoke in the area where the specimens or kits are handled. |
| • | Handle all specimens as if they contain infectious agents. Observe established precautions against microbiological hazards throughout the procedure and follow the standard procedures for proper disposal of specimens. |
| • | Wear protective clothing such as laboratory coats, disposable gloves and eye protection when specimens are assayed. |
| • | Humidity and temperature can adversely affect results. |
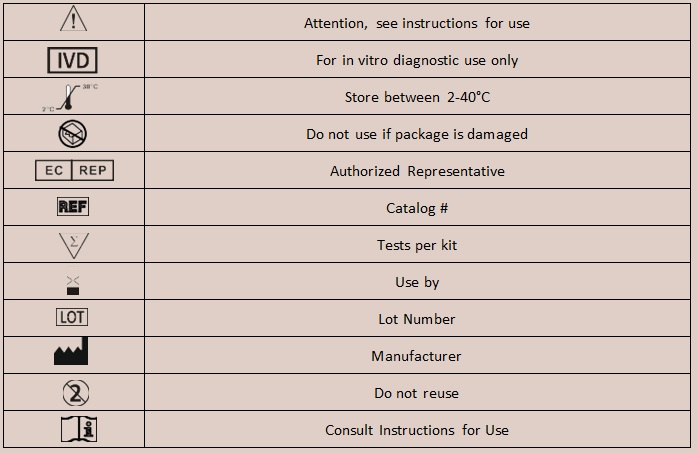
| STORAGE AND STABILITY The kit can be stored at room temperature or refrigerated (2-40°C). The test cassette is stable through the expiration date printed on the sealed pouch. The test cassette must remain in the sealed pouch until use. DO NOT FREEZE. Do not use beyond the expiration date. SPECIMEN COLLECTION AND PREPARATION |
|
| • | The Dengue Combo Rapid Test Cassette (Whole Blood/Serum/Plasma) can be performed using whole blood, serum, or plasma. | |
| • | To collect Fingerstick Whole Blood Specimens: | |
| ⚬ | Wash the patient's hand with soap and warm water or clean with an
alcohol swab. Allow to dry. Massage the hand without touching the puncture site by rubbing down the hand towards the fingertip of the middle or ring finger. Puncture the skin with a sterile lancet. Wipe away the first sign of blood. Gently rub the hand from wrist to palm to finger to form a rounded drop of blood over the puncture site. |
|
| • | Add the Fingerstick Whole Blood specimen to the test cassette by using a 5µl dropper or micropipette measuring 5µl and a 25µl dropper or micropipette measuring 25µl. The 5µl or 25µl droppers provided with the test dispenses approximately 5µl or 25µl in one drop even if more blood is aspirated in the dropper. | |
| • | Separate serum or plasma from blood as soon as possible to avoid hemolysis. Use only clear, non-hemolyzed specimens. | |
| • | Testing should be performed immediately after specimen collection. Do not
leave the specimens at room temperature for prolonged periods. Serum and plasma specimens may be stored at 2-8°C for up to 3 days, for long-term
storage, specimens should be kept below - 20°C. Whole blood collected by venipuncture should be stored at 2-8°C if the test is to be run within
2 days of collection. Do not freeze whole blood specimens. Whole blood collected by fingerstick should be tested immediately. |
|
| • | Bring specimens to room temperature prior to testing. Frozen specimens must be completely thawed and mixed well prior to testing. Specimens should not be frozen and thawed repeatedly. | |
| • | If specimens are to be shipped, they should be packed in compliance
with federal regulations for transportation of etiologic agents. |
|
| MATERIALS |
||
| • Test cassettes | • Droppers | • Buffer | • Package insert |
| • Lancets (for fingerstick whole blood only) | • Alcohol Swabs |
| • Specimen collection containers | • Centrifuge (for plasma only) |
| • Micropipette | • Timer |
|
DIRECTIONS FOR USE Allow the test, specimen, buffer and/or controls to reach room temperature (15-30°C) prior to testing. |
|
| 1. | Bring the pouch to room temperature before opening it. Remove the test cassette from the sealed pouch and use it within 1 hour. |
| 2. | Place the cassette on a clean and level surface. |
| » | For Serum or Plasma specimen: |
| For NS1: | |
| • | To use the dropper: Hold the dropper vertically and transfer 3 drops of serum or plasma (approximately 75µl) to the specimen area, and start the timer. |
| • | To use a micropipette: Pipette 75µl of serum of plasma into the sample well. |
| For IgG/IgM: | |
| • | To use the dropper: Draw up the specimen into the thin dropper tube and add 1 drop of serum or plasma (approximately 5 µl), into the specimen well of the test cassette, then add 1 drop of buffer (approximately 40µl) and start the timer. Avoid trapping air bubbles in the specimen well. |
| • | To use a micropipette: Pipette and dispense 5 µl of serum or plasma to the specimen well of the test cassette, then add 1 drop of buffer (approximately 40 µl) and start the timer. |
| » | For Whole Blood(Venipuncture/Fingerstick) specimen: |
| For NS1: | |
| • | To use the dropper: Hold the dropper vertically and transfer 3 drops of whole blood (approximately 75 µL) to the specimen area, then add 1 drop of buffer (approximately 40 µL) and start the timer. |
| • | To use a micropipette: Transfer 75 µL whole blood specimen to the specimen area of test cassette, then add 1 drop of buffer. |
| For IgG/IgM: | |
| • | To use the dropper: Draw the specimen into the thin dropper tube, and transfer 2 drops of whole blood (approximately 10 µl) to the specimen well of the test cassette, then add 1 drop of buffer (approximately 40 µl) and start the timer. |
| • | To use a micropipette: Dispense 10 µl of whole blood to the specimen well of the test cassette, then add 1 drop of buffer (approximately 40 µl) and start the timer. |
| 3. | Read the results at 10 minutes, do not interpret the results after
20 minutes. |
 INTERPRETATION OF RESULTS IgG and IgM POSITIVE:* Three lines appear. One coloured line should be in the control line region (C), and two coloured lines should appear in IgG test line region and IgM test line region. The color intensities of the lines do not have to match. The result is positive for IgG&IgM antibodies indicated end stage of primary Dengue infection and early stage of secondary Dengue infection. IgG POSITIVE:* Two lines appear. One coloured line should be in the control line region (C), and a coloured line appears in IgG test line region. The result is positive for Dengue virus specific-IgG and is probably indicative of secondary Dengue infection. IgM POSITIVE:* Two lines appear. One coloured line should be in the control line region (C), and a coloured line appears in IgM test line region. The result is positive for Dengue virus specific-IgM antibodies and is indicative of primary Dengue infection. *NOTE: The intensity of the color in the test line region (NS1 and/or IgG and/or IgM) will vary depending on the concentration of Dengue NS1 antigen and/or IgG and/or IgG present in the specimen. Therefore, any shade of red in the test region should be considered positive. NEGATIVE: One colour line appears in the control region (C). No apparent red or pink line appears in the test region (T). INVALID: Control line fails to appear. Insufficient specimen volume or incorrect procedural techniques are the most likely reasons for control line failure. Review the procedure and repeat the test with a new test cassette. If the problem persists, discontinue using the test kit immediately and contact your local distributor. QUALITY CONTROL Internal procedural controls are included in the test. A colour line appearing in the control region (C) is an internal positive procedural control. It confirms sufficient specimen volume and correct procedural technique. Control standards are not supplied with this kit; however, it is recommended that positive and negative controls be tested as a good laboratory practice to confirm the test procedure and to verify proper test performance. |
| LIMITATIONS | |
| 1. | The Dengue Combo Rapid Test Cassette (Whole Blood/Serum/Plasma) will only indicate the presence of Dengue NS1 antigen and Dengue antibodies in the specimen and should not be used as the sole criteria for the diagnosis of Dengue. |
| 2. | The Assay Procedure and the Assay Result Interpretation must be followed closely when testing the presence of dengue Ag in serum or plasma from individual subjects. Failure to follow the procedure may give inaccurate results. |
| 3. | A negative test result for Dengue NS1 does not preclude the possibility of exposure to or infection with dengue viruses. |
| 4. | A negative result for Dengue NS1 can occur if the quantity of dengue Ag present in the specimen is below the detection limits of the assay, or the dengue Ag that are detected are not present during the stage of disease in which a sample is collected. |
| 5. | Some specimens containing unusually high titer of heterophile antibodies or rheumatoid factor may affect expected results. |
| 6. | If the symptom persists, while the result from Dengue NS1 Rapid Test is negative or non-reactive result, it is recommended to re-sample the patient few days late or test with an alternative test device such as PCR, ELISA. |
| 7. | The results obtained with this test should only be interpreted in conjunction with other diagnostic procedures and clinical findings. |
| 8. | In the early onset of fever, anti-Dengue IgM concentrations may be below detectable levels. For primary infection, an IgM antibody-capture enzyme-linked immune sorbent assay (MACELISA) showed that 80% of the Dengue patients tested exhibited detectable levels of IgM antibody by the fifth day after infection, and 99% of the patients tested IgM positive by day 10. It is recommended that patients be tested within this time. For the secondary infection, a low molar fraction of anti-Dengue IgM and a high molar fraction of IgG that is broadly reactive to flaviviruses characterize the antibodies.5 The IgM signal may be faint and the cross reaction in the region of IgG line may appear. |
| 9. | Serological cross-reactivity across the flavivirus group (Dengue 1, 2, 3 & 4, St. Louis encephalitis, West Nile virus, Japanese encephalitis and yellow fever viruses) is common.6,7,8 Positive results should be confirmed by other means. |
| 10. | The continued presence or absence of antibodies cannot be used to determine the success or failure of therapy. | 11. | Results from immunosuppressed patients should be interpreted with caution. |
| 12. | As with all diagnostic tests, all results must be interpreted together with other clinical information available to the physician. | 13. | If the test result is negative and clinical symptoms persist, additional
testing using other clinical methods is recommended. A negative result does not at any time preclude the possibility of Dengue infection. |
| EXPECTED VALUES The Dengue NS1 Rapid Test Cassette (Whole Blood/Serum/Plasma) has been compared with a leading commercial Dengue Ag EIA test. The correlation between these two systems is 96.0%. Primary Dengue infection is characterized by the presence of detectable IgM antibodies 3-5 days after the onset of infection. Secondary Dengue infection is characterized by the elevation of Dengue- specific IgG. In the majority of the cases, this is accompanied by elevated levels of IgM. The Dengue Rapid Test Cassette (Whole Blood/Serum/Plasma) has been compared with a leading commercial Dengue ELISA test, demonstrating sensitivity of 83.3% for IgM in primary infection and 98.4% for IgG in secondary infection. PERFORMANCE CHARACTERISTICS The results show that the relative sensitivity of the Dengue NS1 Rapid Test Cassette (Whole Blood/Serum/Plasma) is 95.8%, and the relative specificity is 96.1%.And the overall relative sensitivity for the primary and secondary infection of the Dengue Rapid Test Cassette (Whole Blood/Serum/Plasma) is 94.3%, and the relative specificity is 99.1%, and the relative accuracy is 98.3%. Dengue IgG/IgM  Relative sensitivity: (20+63)/ (24+64) =94.3% (95%CI*: 87.2%~98.1%); Relative specificity: 429/433=99.1% (95%CI*: 97.7%~99.7%); Accuracy: (20+63+429)/ (24+64+433) =98.3% (95%CI*: 96.7%~99.2%). *Confidence Intervals  Relative sensitivity: 137/143*100%=95.8% (95%CI*: 91.1%~98.4%); Relative specificity: 200/208*100%=96.1% (95%CI*: 92.6%~98.4%); Accuracy: (137+200)/(137+6+8+200)*100%=96.0%(95%CI*:93.4%~97.8%) *Confidence Intervals Intra-Assay Acetaminophen: 20 mg/dL Acetylsalicylic Acid: 20 mg/dL Ascorbic Acid: 2g/dL Bilirubin: 1g/dL Creatin: 200 mg/dL Caffeine: 20 mg/dL Gentisic Acid: 20 mg/dL Hemoglobin 1000mg/dL Albumin: 2 g/dL Oxalic Acid: 60mg/dL None of the substances at the concentration tested interfered in the assay. BIBLIOGRAPHY |
|
| 1. | Halstead SB, Selective primary health care: strategies for control of disease in the developing world: XI, Dengue. Rev. Infect. Dis. 1984; 6:251-264. |
| 2. | Halstead SB, Pathogenesis of dengue: challenges to molecular biology. Science 1988; 239:476-481. |
| 3. | Ruechusatsawat K, et al. Daily observation of antibody levels among dengue patientsdetected by enzyme-linked immunosorbent assay (ELISA).Japanese J. Trop. Med.Hygiene 1994; 22: 9-12 |
| 4. | Lam SK. Dengue haemorrhagic fever. Rev. Med. Micro. 1995; 6:39-48 |
| 5. | Dengue haemorrhagic fever: diagnosis, treatment, prevention and control. 2nd edition.Geneva: World Health Organization |
| 6. | Yamada K, et al. Antibody responses determined for Japanese dengue fever patients by neutralization and hemagglutination inhibition assays demonstrate cross-reactivity between dengue and Japanese encephalitis viruses. Clin Diagn Lab Immunol. 2003 Jul; 10(4): 725-8. |
| 7. | Dobler G, et al. Cross reactions of patients with acute dengue fever to tick-borne encephalitis. Wien Med Wochenschr (in German). 1997; 147(19-20): 463-4 |
| 8. | Makino Y, et al. Studies on serological cross-reaction in sequential
flavivirus Microbiol Immunol. 1994; 38(12): 951-5. |
 |
|
 |
ICT INTERNATIONAL 271 De Goed Hoop Estate Noordhoek Cape Town South Africa www.ictdiagnostics.co.za Tel: +27 82 441 1922 Email: russellg@icon.co.za |