Cassette (Faeces) Package Insert REF CHOL01 - English A rapid test for the qualitative detection of Vibrio cholerae O1 and Vibrio cholerae O139 in the specimens of human faeces.For professional in vitro diagnostic use only. [INTENDED USE] The Vibrio choleraeO1/O139 Combo Rapid Test Cassette (Faeces) is a rapid chromatographic immunoassay for the qualitative detection of Vibrio cholerae O1 and Vibrio cholerae O139 in human feces to aid in the diagnosis of Vibrio cholerae O1 or Vibrio cholerae O139 infection. [SUMMARY] cholerae is an acute watery diarrheal disease caused mainly by Vibrio cholerae serogroup O1 and less commonly by V. cholerae O139. cholerae can lead to severe diarrhea and death if untreated. V. cholerae O1 and V. cholerae O139 are transmitted through fecal-oral contamination, and cholera is thus predominantly associated with lack of safe drinking water, proper sanitation and personal hygiene. cholerae is an important public health problem in many parts of Asia, Africa and Latin America [1, 2]. Globally, 3-5 million cases and over 100,000 deaths occur annually due to cholera [3]. Countries facing complex emergencies are more vulnerable to cholera outbreaks [4]. The etiological agent of cholera has been identified as Vibrio cholerae (V. cholerae), a gram negative bacterium, which is generally transmitted to humans via contaminated water and food. The species V. cholerae is divided into several serogroups on the basis of O antigens. The subgroups O1 and O139 are of special interest because both can cause epidemic and pandemic cholera. It is critical to determine as quickly as possible the presence of V. cholerae O1 and O139 in clinical specimens, water, and food so that appropriate monitoring and effective preventive measures can be undertaken by public health authorities. The Vibrio cholerae O1/O139 Combo Rapid Test Cassette (Faeces) is a rapid chromatographic immunoassay for the qualitative detection of Vibrio cholerae O1 and Vibrio cholerae O139 in human faeces, providing results in 10 minutes. The test utilizes antibodies specific for VC O1 and VC O139 antigens to selectively detect VC O1 antigens and VC O139 antigens in human faeces. [PRINCIPLE] The Vibrio cholerae O1/O139 Combo Rapid Test Cassette (Faeces) is a qualitative, lateral flow immunoassay for the detection of Vibrio cholerae O1 and Vibrio cholerae O139 antigens in human faeces. In Vibrio cholerae O1 test, the membrane is pre-coated with anti-Vibrio cholerae O1 antibody on the test line region of the test. During testing, the specimen reacts with the particle coated with anti-Vibrio cholerae O1 antibody. The mixture migrates upward on the membrane by capillary action to react with anti-Vibrio cholerae O1 antibodies on the membrane and generate a coloured line. In Vibrio cholerae O139 test, the membrane is pre-coated with anti-Vibrio cholerae O139 antibody on the test line region of the test. During testing, the specimen reacts with the particle coated with anti-Vibrio cholerae O139 antibody. The mixture migrates upward on the membrane by capillary action to react with anti-Vibrio cholerae O139 antibodies on the membrane and generate a coloured line. The presence of this coloured line in the test region indicates a positive result, while its absence indicates a negative result. To serve as a procedural control, a coloured line will always appear in the control line region indicating that proper volume of specimen has been added and membrane wicking has occurred. [REAGENTS] The test cassette contains monoclonal anti-Vibrio cholerae O1 antibody coated particles and monoclonal anti-Vibrio cholerae O1 antibody coated on the membrane. The test cassette contains monoclonal anti-Vibrio cholerae O139 antibody coated particles and monoclonal anti-Vibrio cholerae O139 antibody coated on the membrane. [PRECAUTIONS] |
| * | For professional in vitro diagnostic use only. Do not use after the expiration date. | ||
| * | The test should be remained in the sealed pouch until use. | ||
| * | Do not eat, drink or smoke in the area where the specimens or kits are handled. | ||
| * | Handle all specimens as if they contain infectious agents. Observe established precautions againstmicrobiological hazards throughout all procedures and follow the standard procedures for proper disposal of specimens. | ||
| * | Wear protective clothing such as laboratory coats, disposable gloves and eye protection when specimens are assayed. | ||
| * | The used test should be discarded according to local regulations. | ||
| * | Humidity and temperature can adversely affect results. |
||
|
[MATERIALS ] Materials provided • Test cassettes • Specimen Collection Tubes with extraction buffer • Package insert • Droppers Materials required but not provided • Specimen collection containers • Pipette and Disposable tips (optional) • Centrifuge • Timer |
|||
| [STORAGE AND STABILITY] The kit can be stored at room temperature or refrigerated (2-30°C). The test cassette is stable through the expiration date printed on the sealed pouch. The test cassette must be remained in the sealed pouch until use. DO NOT FREEZE. Do not use beyond the expiration date. [SPECIMEN COLLECTION AND PREPARATION] |
|||
| * | The fecal specimen must be collected in clean, dry, waterproof container containing no detergents,preservatives or transport media. | ||
| * | Bring the necessary reagents to room temperature before use. | ||
| * | Store the specimens at
-20°C if the specimens cannot be tested in 3 days. |
||
| [DIRECTIONS FOR USE] Allow the test cassette, specimen, buffer and/or controls to reach room temperature (15-30°C) prior to testing. |
|
| For Fecal Specimens: |
|
| 1. | To collect faecal specimens: Collect sufficient quantity of feces (1-2 mL or 1-2 g) in a clean, dry specimen collection container to obtain maximum antigens (if present). Best results will be obtained if the assay is performed within 6 hours after collection. Specimen collected may be stored for 3 days at 2-8°C if not tested within 6 hours. For long term storage, specimens should be kept below -20°C. |
| 2. | To process faecal specimens:
• For Solid Specimens: Unscrew the cap of the specimen collection tube, then randomly stab the specimen collection applicator into the faecal specimen in at least 3 different sites to collect approximately 50 mg of faeces (equivalent to 1/4 of a pea). Do not scoop the faecal specimen. • For Liquid Specimens: Hold the dropper vertically, aspirate fecal specimens, and then transfer 2 drops of the liquid faeces (approximately 80 µL) into the specimen collection tube containing the extraction buffer. • Tighten the cap onto the specimen collection tube, and then shake the specimen collection tubevigorously to mix the specimen and the extraction buffer. Leave the tube alone for 2 minutes. |
| 3. | Bring the pouch to room temperature before opening it. Remove the test cassette
from the foil pouch and use it within one hour. Best results will be obtained if the test is performed immediately after opening the foil pouch. |
| 4. | Hold the specimen collection tube upright and open the cap onto the specimen
collection tube. Invert the specimen collection tube and transfer 2 full drops of the extracted specimen (approximately 80µL) to each specimen well
(S) of the test cassette, then start the timer. Avoid trapping air bubbles in the specimen well (S). See illustration below. |
| 5. | Read results at 10 minutes after dispensing the specimen. Do not read results
after 20 minutes. |
| Note: If the specimen does not migrate (presence of particles),
centrifuge the extracted specimenscontained in the extraction buffer vial. Collect 80 µL of supernatant, dispense into the specimen well (S) of a new
test cassette and start afresh following the instructions mentioned above. |
 [INTERPRETATION OF RESULTS] *NOTE: The intensity of the colour in the test line region (T) will vary depending on the concentration of Vibrio cholerae O1 antigen present in the specimen. Therefore, any shade of color in the test line region (T) should be considered positive. |
| VC O139 POSITIVE:* Two lines appear in VC O139 window. One colorued line should be in
the control line region (C) and another apparent coloured line should be in the test line region (T). *NOTE: The intensity of the color in the test line region (T) will vary depending on the concentration of Vibrio cholerae O139 antigen present in the specimen. Therefore, any shade of colour in the test line region (T) should be considered positive. |
|
| *NEGATIVE: One coloured line appears in the control line region (C).
No line appears in the test line region. (T). INVALID: Control line fails to appear. Insufficient specimen volume or incorrect procedural techniques are the most likely reasons for control line failure. Review the procedure and repeat the test with a new test. If the problem persists, discontinue using the test kit immediately and contact your local distributor. [QUALITY CONTROL] Internal procedural controls are included in the test. A coloured line appearing in the control region (C) is an internal valid procedural control. It confirms sufficient specimen volume and correct procedural technique. Control standards are not supplied with this kit; however, it is recommended that positive and negative controls be tested as a good laboratory practice to confirm the test procedure and to verify proper test performance. [LIMITATIONS] |
|
| 1. | The Vibrio cholerae O1/O139 Combo Test Cassette (Faeces) is for in vitro diagnostic use only. The test should be used for the detection of Vibrio cholerae O1 antigens and Vibrio cholerae O139 antigens in feces. Neither the quantitative value nor the rate of increase in Vibrio cholerae O1 antigens and Vibrio cholerae O139 antigens concentration can be determined by this qualitative test. |
| 2. | As with all diagnostic tests, all results must be interpreted together with other clinical information available to the physician. |
| 3. | If the test result is negative and clinical symptoms persist, additional testing using other clinical methods is recommended. A negative result does not at any time preclude the possibility of Vibrio cholerae O1 or Vibrio cholerae O139 infection. |
| 4. | Following certain antibiotic treatments, the concentration of Vibrio cholerae O1 antigens and Vibrio cholerae O139 antigens may decrease to the concentration below the minimum detection level of the test. Therefore, diagnosis should be made with caution during antibiotic treatment. |
[EXPECTED VALUES] The Vibrio cholerae O1/O139 Combo Rapid Test Cassette (Faeces) has been compared with PCR methods, demonstrating an overall accuracy of 97%. [PERFORMANCE CHARACTERISTICS] For Vibrio cholerae O1 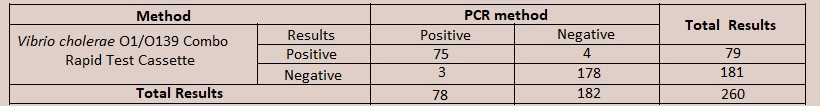 Relative Specificity: 97.8% (95%CI*: 94.5%-99.4%) Overall Accuracy: 97.3% (95%CI*: 94.5%-98.9%) For Vibrio cholerae O139 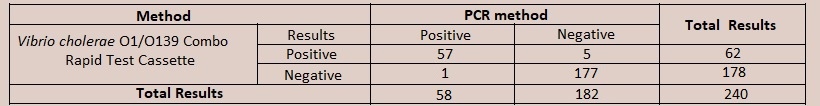 Relative Specificity: 97.3% (95%CI*: 93.7%-99.1%) Overall Accuracy: 97.5% (95%CI*: 94.6%-99.1%) [NOTE] The Vibrio cholerae O1/O139 Combo Rapid Test Cassette (Faeces) can also be used for detection of V. cholerae O1 and V. cholerae O139 antigens in environmental water. For this, environmental water should be collected in a clean and dry container and test should be performed by transfering 2 drops of environment water (approximately 80µL) to the specimen well. Test time is 10 minutes same as fecal specimens. For Interpretation of results also, the interpretation is same as with faecal specimen. Testing with environmental water is based on the technical premises that V. cholerae O1 and V. cholerae O139 antigens are available in environmental water same as in fecal specimen.  Intra-Assay |
|
| Citrobacter freundii | Clostridium difficile | Coxsackie |
| Candida albicans | Chlamydia trachomatis | Echovirus |
| Enterococcus faeciumz | E.coli | Enterococcus faecalis |
| Gardnerella vaginalis | Neisseria gonorrhea | Proteus mirabilis |
| Proteus vulgaris | Pseudomonas aeruginosa | Staphylococcus aureus |
| Adenovirus | Shigella dysenteriae | Shigella flexneri |
| Corynebacterium diphtheria |
||
|
[BIBLIOGRAPHY] |
|
| 1. | Khazaei HA, Rezaei N, Bagheri GR, Moin AA. A six-year study on Vibrio cholerae in southeastern Iran.Japanese journal of infectious diseases. 2005; 58(1):8-10. |
| 2. | Shears P. Recent developments in cholera. Current opinion in infectious diseases. 2001; 14(5):553-8. |
| 3. | Cholerae vaccines: WHO position paper. Wkly Epidemiol Rec. 2010; 85(13):117-28. |
| 4. | Organization WH. Prevention and control of cholera outbreaks: WHO policy and
recommendations.Geneva: World Health Organization, Global Task Force on cholerae Control. 2010. |
 ICT DIAGNOSTICS 271 De Goede Hoop Estate Village Lane Noordhoek South Africa Tel: +27 82 441 1922 Email : russell@ictdiagnostics.co.za www.ictdiagnostics.co.za Number: 145908800 Effective date:2023-05-30 |
|